What you can build on Uscreen
Take control of your audience & brand.
End the revenue rollercoaster.
Turn your fans into loyal members on your own membership platform across all devices.
Own your own brand so you can live life on your own terms.
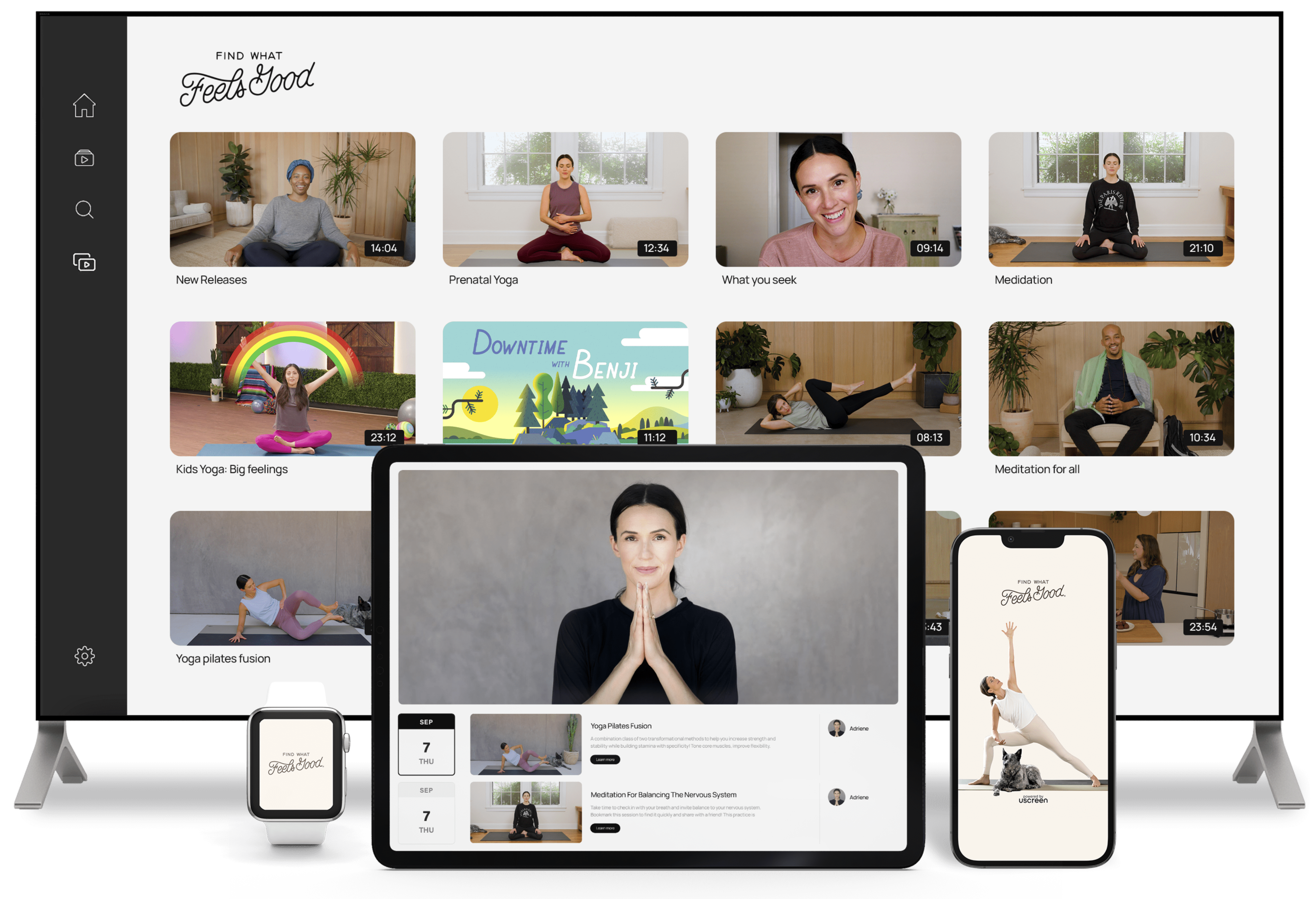
The only membership platform where
content and community seamlessly unite
Our platform is built around what is proven to engage, retains and grow your audience.
Imagine a Netflix-style video library, a safe community space, and direct livestreams,
all under your white-labeled apps and website.
$100K+
Monthly revenue
2x
membership growth
4.5⭑
average app store rating
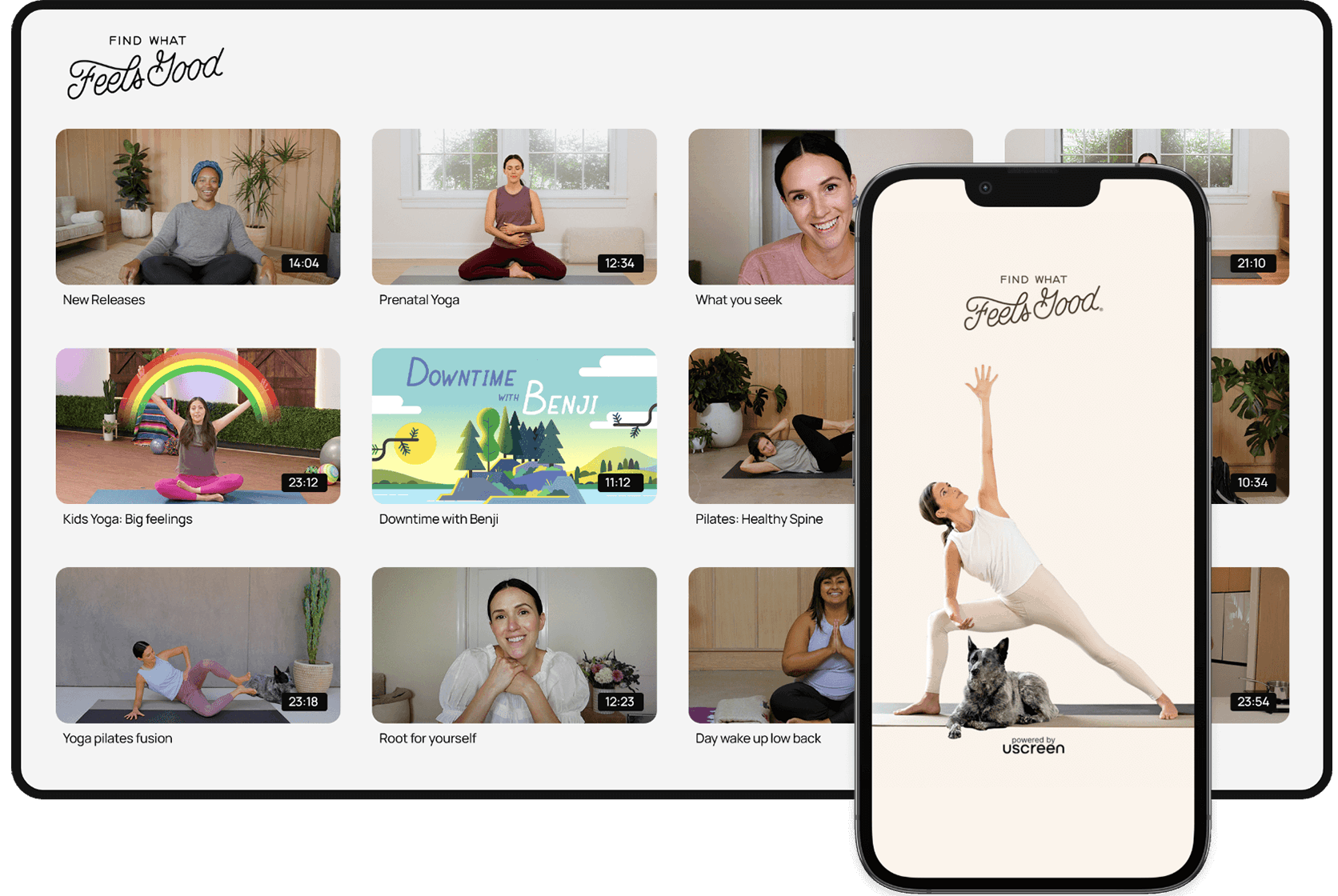
Engage with a video experience inspired by Netflix & YouTube
Showcase your content through our user-friendly interface designed for easy discovery and binge-watching. As members enjoy your videos, they can effortlessly comment, download, and curate their own playlists.
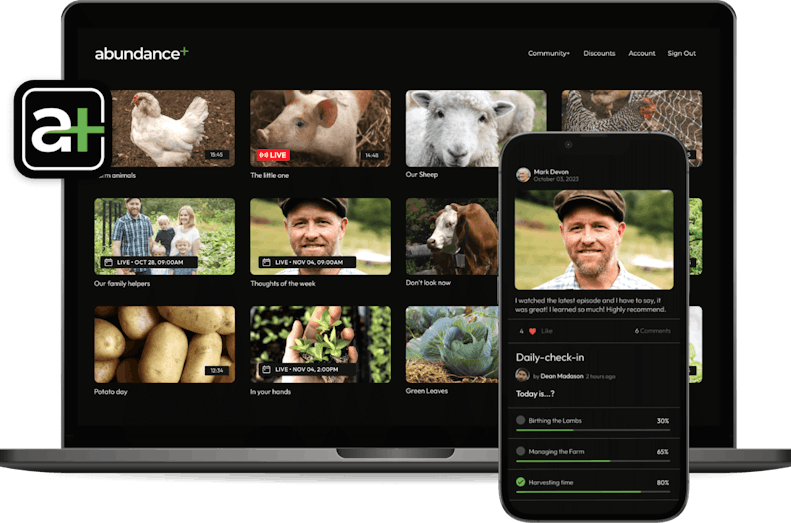
Build a supportive community and create a self-sustaining ecosystem
Draw in and keep members with a safe space, right next to your video library, always within their reach. Sustain your community as you enable members to engage, inspire, and connect with each other.

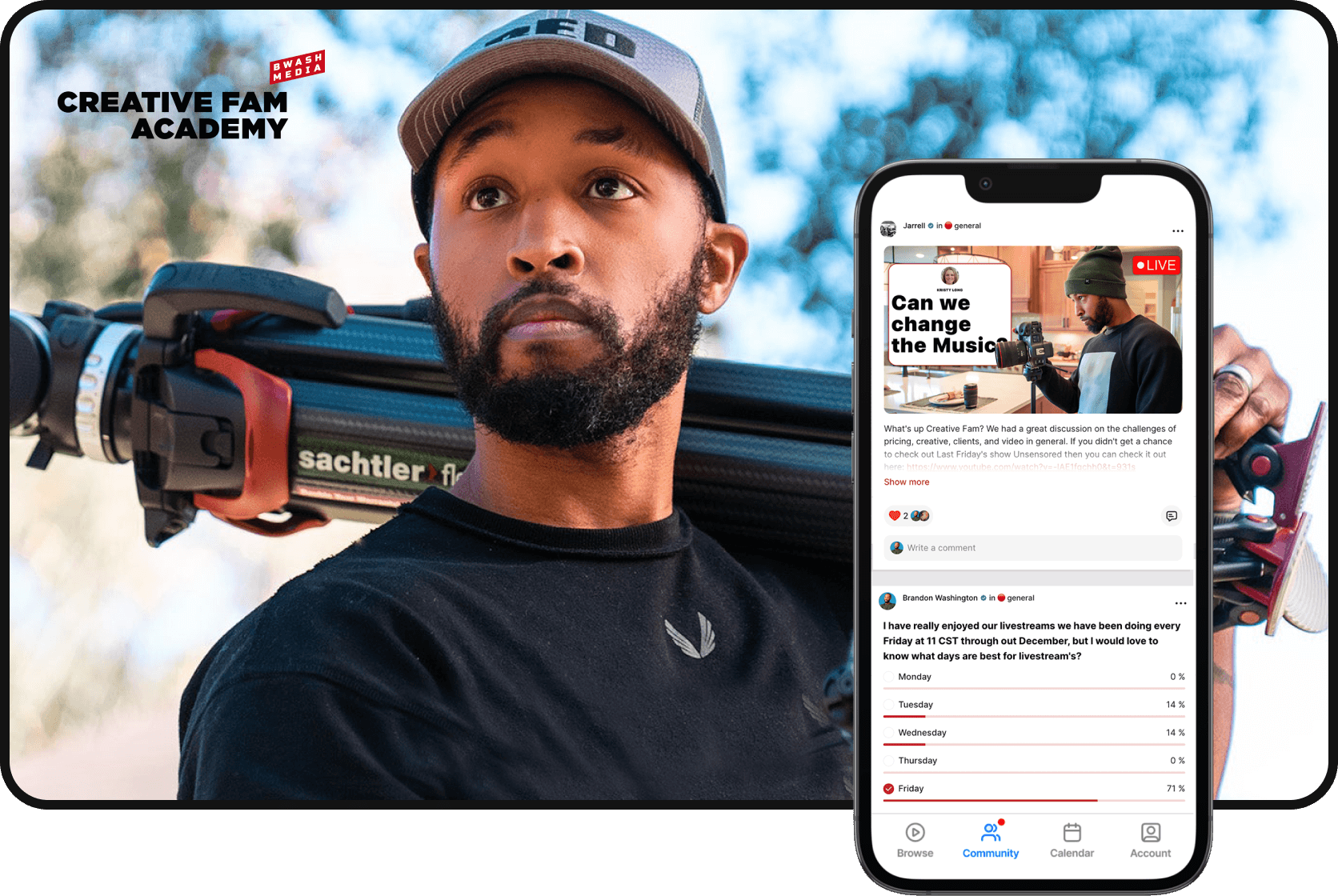
Make your community
a key part of your membership
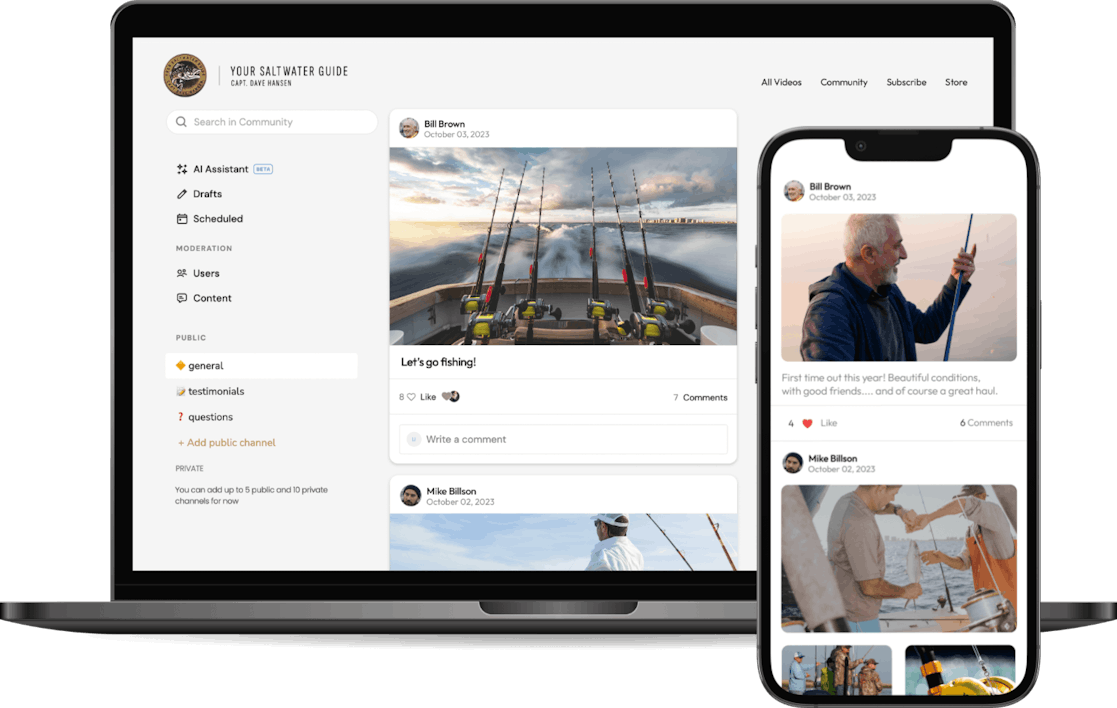
Let your fans connect with each other with our Community feature built right into your membership. With public and private channels and interactive community features, a community will make your membership even more valuable for your fans.
We could be gigantic - we haven’t even touched the surface of it, and we’re not slowing down. And it's the personal touch and the community that's going to drive this thing.
Captain Dave Hansen, Founder
Your Saltwater Guide
Everything you need for an amazing membership
1:1 support every step of the way
You won’t be alone on your membership journey - we will walk you through setting up your business model, content strategy, pricing and everything else you need to make your membership a huge success.
Flexible monetization tools
Design your subscription offering however you want, including free trials, one-off courses and a variety of payment options. Your membership will have all the best practices built right in.
We partner with you every step of the way
We go beyond being a technology platform. We’ll help design your membership
and provide dedicated expert onboarding and account managers.
We make your success our success.


Having a lot of help with the marketing was great. It’s very intuitive, easy to use and I could get it up and running quickly.
Tom Gellie
Big Picture Skiiing
$33K+
Monthly revenue
950+
paid subscribers
66%
increase in revenue
Free, fast and easy migrations
Thousands of creators have had seamless transitions to Uscreen thanks to our expert Migrations team. We’ll take care of all the hard work, free of charge.
A team of memberships experts has your back
Work with your very own Success Manager for a worry-free journey. Launch and manage your membership effortlessly with Uscreen’s friendly, responsive team in your back pocket.
24/7 support for you and your members
Seamless issue resolution and instant answers to all your inquiries with our speedy customer support, for both you and your end users. Leave the boring, painful stuff to us so you can focus on what you do best—creating content.
The market’s first community for membership operators
Get strategies, tips, and case studies specifically for membership success. Connect with top creators and exchange best practices in our community.
Trusted by 4,000+ Video Creators
Unlike other platforms that support various content formats and monetization options,
Uscreen is built around what is proven to work for video content memberships.
Don't just take our word for it
Explore authentic reviews from customers subscribed to membership platforms powered by Uscreen.